You may have thought you left physics and chemistry behind when you left school, but if you want to eat well, you need to understand that cooking is all about physics and chemistry, with a little magic mixed in. Here are some foundational concepts every outdoor cook needs to know.
What is cooking? Foods are composed mainly of water, fat, protein, and carbohydrates. Cooking is the process of changing the chemistry of food usually by transferring energy to the food long enough so that it is safe and digestible, and achieves the desired flavor, texture, tenderness, juiciness, appearance, and nutrition. It is really really important to stop thinking about heat or temperature when you talk about cooking and start thinking about energy. They are related, but different.
The three ways energy cooks food outdoors. Food gets hot when molecules vibrate so fast that their temperature rises. When cooking outdoors, energy is transferred to food by three methods. Which one you use is crucial. These processes have been described this way:
- Conduction is when your lover’s body is pressed against yours.
- Convection is when your lover blows in your ear.
- Radiation is when you feel the heat of your lover’s body under the covers without touching.
Let’s be a bit more precise:
1) Conduction is when energy is transferred to the food by direct contact with the eneregy source. Grill marks are a good example of conduction. Heat is transferred to the grill grates and the hot metal brands the meat. Although the air and the grates measure the same temperature, the grates store and transmit more energy that the air. That’s why there are stripes on the hot dog.
2) Convection is when energy is carried to the food by a fluid. Air, water, and oil are all fluids. The hot dog not touching the metal grates is surrounded by hot air and that is convection cooking. So is boiling it in water, or deep frying it in oil (you really need to try a deep fried hot dog once). If you get one side of your grill hot and put the food on the other side (2-zone cooking), it is cooked by natural convection airflow. Most gas grills cook mostly by convection. A conventional indoor oven uses natural convection airflow to cook, but a “convection oven” and an “air fryer” have a fan and an extra heat source near the fan and uses forced airflow. A convection oven cooks 25% to 30% faster. It’s like wind chill in reverse. The moving warm air blows away the cold air bubble surrounding the cold food and transfers more energy than stagnant warm air. In either case, airflow only cooks the exterior of the meat, the interior of the meat is cooked by conduction as the energy builds up on the surface and then travels through the meat.
3) Thermal Radiation is the transfer of heat by direct exposure to a source of light energy, a.k.a. infrared (IR) energy. Put bread in your toaster and it cooks by thermal radiation. Put a hot dog on a coat hangar and hold it to the side of a campfire and you are cooking by thermal radiation. IR is how most charcoal grills cook. Think of it like sunburn. There’s more IR in standing in the sun than ultraviolet (UV). If you want a dark brown sear on a steak, and you do want the flavors created by searing, you need IR.
It is really really important to understand the differences between these energy sources. When people brag that theur Big Green Avocado gets up to 600°F: It. Just. Doesn’t. Matter. They are talking strictly about the convection temp and warm air just can’t sear a steak like radiation. Besides, you want to cook most foods at a low temp, and when you want to sear it you want direct concentrated IR more than air temp. That’s why a $30 Hibachi will sear a steak better than a $1,000 Big Green Avocado.
To be complete, I need to mention that indoors there are two other methods heat is applied for food:
4) Excitation is how microwave ovens work. Microwaves are radio waves that penetrate the food and vibrate the molecules inside the hot dog until it gets hot without heating the air around it. Water in the food heats first in the microwave. Tis is another form of radiation. As yet, I haven’t seen a grill that cooks with microwaves.
5) Induction is the latest technique for stove tops. A copper coil is placed under a smooth cooktop and an alternating current is sent through the coil creating a rapidly changing electromagnetic field. Electrons in steel or cast iron pots on the cooktop above the electromagnet are jostled by the rapidly changing magnetism, but they resist and the resistance heats the pot. The pot then transfers the heat by conduction to the food without the cooktop or the air around it getting hot. Induction is the most efficient way to cook, and it very responsive to the control knob, but it does not work with aluminum, glass, or copper pots.
The difference between heat and temperature
Put an empty frying pan and pot of water in a 200°F oven for an hour. Open the door and stick your hand in the warm air of the oven and count how long you can hold your hand in there. Perhaps a minute? Then plunge one hand in the 200°F water and place the other hand on the frying pan. After you get back from the hospital, ponder the fact that not everything that is 200°F transfers energy at the same rate.
Cooking by convection air in a hot oven is a lot slower than by conduction from a pan because steel is 8,000 times more dense (it has more molecules in a cubic inch) than air which conducts heat at 1/50 the rate of steel. You can speed the process a bit by adding a fan to the oven and moving the air around as is done in a “convection oven”, but all ovens are really convection ovens. Water is more dense than air and it holds more energy so it cooks faster than in the oven, and oil cooks faster still.
According to the AmazingRibs.com science advisor, Prof. Greg Blonder, “Temperature measures the average energy of each atom. Heat is the total for all atoms. It’s like money. If the average income in the US is $40,000 per family, the total wages are in the trillions.”
Also, radiant heat delivers more energy than convection heat. Let’s say you have two gas grills. On one grill, the two burners on the right side are on full and two burners on the left are turned off. The air temp on the left side, the indirect heat side, might be 325°F as convection flow of air from the right side circulates over the left side. Let’s put a steak and a big honkin prime rib roast on the left, indirect side.
On the second grill you have all four burners on medium and the air temp on the both sides is also 325°F. Let’s put a steak and a roast on this grill too. The steak’s bottom will brown better on the second grill because it is above direct radiant heat which is imparting more energy than the first grill where the heat is from convection air. But by the time the roast is done, it will be blacker than a mourning hat.

On a charcoal grill, you can increase energy simply by increasing the number of coals. The distance from the energy source is also an important factor. Energy dissipates as you move away from the heat source. In a $800 grill made of ceramics the heat source may be 2′ from the cooking surface while on an $89 Weber Kettle the charcoal is only 4″ away, and on a $30 Hibachi the coals may be as little as 1″ away. The walls of a ceramic cooker are thick, they absorb huge amounts of heat, and then release it evenly making a very efficient oven with very steady cooking temps and very low fuel use. Perfect for roasts. But a steak on the cooking surface will not brown as well as on the Weber or even the Hibachi because the coals are so far away and because much of the heat comes from the ceramic side walls and dome, not glowing radiant hot coals right below the meat.
When is food ready to eat?
Food is ready to eat when it is safe and the target temperature is reached. A medium rare steak is 130 to 135°F in the center regardless of how thick, how much it weighs, or what temperature it is cooked at. The single best way to tell if it is time to eat is with an accurate digital thermometer. You cannot tell by poking the meat unless you’re a pro who has cooked the same steaks on the same grill for years. The single thing you can do to improve your cooking is to get a good digital instant read meat thermometer. The second most important thing you can do is get a good digital oven thermometer.
OK, some foods are hard to read with thermometers. You don’t need a thermometer to tell when asparagus is ready, and because of all the bones it is almost impossible to tell when ribs are ready. But for most meats, there is no substitute for knowing precisely what temp the food is.
How heat moves within meat
The most important thing to understand is that when you put food in an enclosed vessel like a grill, smoker, or an oven, the hot air, which has lots of vibrating molecules, transfers some of its energy to the exterior of the meat. The energy in the air excites the molecules on the surface of the meat and then they transfer heat to the molecules inside it and so on, slowly passing the energy towards the center of the meat like a wave of heat. It takes time because meat is about 75% water, and water is a good insulator, especially when trapped inside muscle. The heat moves inward because physics dictates the the meat seeks equilibrium in an effort to make the temperature the same from edge to edge. So most of the meat is cooked by the meat, not by the air.
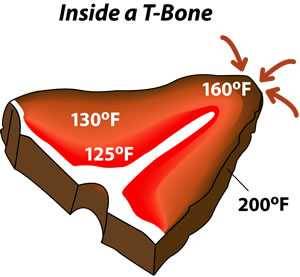
Heat also cooks points and corners more because it can attack on multiple fronts. That’s why the brownies and lasagna in the corners are crunchier. And bones heat at a different rate than muscle tissue because they are filled with air or fat, so in most cases they warm more slowly than the rest of the meat. Click here to read more about the effect bones have on cooking and have some myths debunked.
When you remove the meat from the heat, it continues to seek equilibrium and continues to cook because the heat built up in the outer layers of the meat continues to be passed down towards the center while some is escaping into the air and cooling the exterior. This phenomenon is called carryover cooking.
A thick piece of meat such as a turkey breast might rise as much as 10°F in about 15 minutes after removing it from the grill because of carryover. A thinner piece of meat such as a thick steak might only rise a couple of degrees, and chicken breast may not rise at all. This is important to know this because 5 to 10°F can make the difference between moist turkey and cardboard, a medium rare steak and a drier medium steak. To compensate, use a good digital thermometer and remove thick cuts of meat about 5°F below your target temp.
Thickness is more important than weight
The temperature of your food moves slowly upward during cooking, but the thickness of the meat is a major factor in how long it takes to get the center to the desired temperature. A thin steak cooks faster than a thick steak. And a 5″ thick prime rib that weighs 8 pounds will be done in the same time as a 5″ thick prime rib that weighs 12 pounds. So any recipe that says “cook your steak for three minutes per side” without specifying the thickness of the steak, is seriously flawed. Likewise any recipe that says “cook your roast 30 minutes per pound” is suspect.
About boiling temps and how they impact cooking
Another important concept to know is that when liquid is heated, its temp will increase until it hits its boiling temp and not go any higher. So no matter how high we turn the burners under a pot of water, the water will not get hotter than 212°F (at sea level). If we have a sauce made with water and olive oil it will heat to the boiling temp of water until all the water boils off, and then rise to the boiling temp of the oil, which is usually over 500°F depending on the type of oil. Usually, before oil boils, it starts to smoke, and then it bursts into flames.
This phenomenon comes into play in many aspects of cooking. When you simmer or boil a home made barbecue sauce, because the water cannot exceed 212°F, you warm everything evenly without burning the ingredients (if you stir it so it doesn’t stick to the bottom and dry out). If you simmer potatoes the starches on the inside get soft and the outside doesn’t burn as it might on a grill.
Steam can form at a lower temperature than 212°F as molecules of water get hot and escape the surface as the water warms. That’s why we see water vapor escaping a pot of water before it boils. When water boils, the steam can be hotter than 212°F if it comes into contact with the walls of a hot pot or if it is under pressure, but food submerged in boiling water will cook faster than food bathed in steam because under water the food’s surface is completely surrounded by hot water molecules and in steam there is a lot of air mixed in and air does not conduct heat as well as water.
Because meat is about 75% water, it is useful to think of it as if it is a saturated sponge. As it heats, water on the surface is energized and some of it escapes as steam. Even though the oven may be a lot hotter than 212°F, the surface will stick at close to 212°F as more and more water steams away. As the hotter molecules escape the cooler ones are left behind so the temperature of the meat is cools until water stops evaporating, usually because the surface dries out making a crust or bark. At low cooking temperatures such as 225°F, the temperature I recommend for a lot of my recipes, the rate of evaporation can be so great that the surface and the interior of the meat can stick, usually in the 150 to 165°F range and remain pretty much unchanged for hours, totally freaking out the cook. This phenomenon, called the stall, does not happen if the oven temp is higher, say 325°F, another temperature I recommend frequently.
The stall has four benefits: (1) It helps create the bark which can be very tasty, (2) it holds the meat at a temperature where fats and connective tissues can liquefy, significantly improving texture, juiciness, and flavor, (3) it gives enzymes a chance to tenderize the meat, and (4) it permits the meat to heat evenly so the center and exterior are similar in temperature.
If you soak wood chips in water, as many books recommend, first of all they won’t absorb much (that’s why boats are built from wood), but when you toss them on the coals, the wood stays a cool 212°F until the water boils off. It cannot smoke until it hits a much higher temp, so soaking wood chips so they will smolder better is just another old husband’s tale. Click here to read more on soaking wood.
One other thing about the boiling point: It changes as you move up or down from sea level. At sea level, a pot of water on a flame has a column of air sitting on top of it that weight 14.7 pounds per square inch (psi). At 5,000 feet it’s only 12.3 psi, so water vapor has less resistance when it tries to escape. As a result, the boiling point is lower and leavening gases in breads and cakes expand more quickly. The impact on grilling or smoking is minimal, although there can be higher humidity in a smoker during a long cook and that can cool the meat slowing the process, but not a lot, and it can be offset because natural humidity can be lower at altitude. But altitude can have a profound effect on braising and other wet cooking methods. It can even impact slow cookers, microwaves, and frying.

The magic of infrared radiation
Infrared radiation is the best method for delivering high heat to food on a grill. Charcoal grills produce a lot of direct scorching infrared radiation that is converted to heat when it strikes food. Gas and pellet grills produce mostly convection heat.

If you remember your high school science, you know that infrared is a section of the wavelength continuum around us, just up the road from visible light and down the road from the radio in your car.
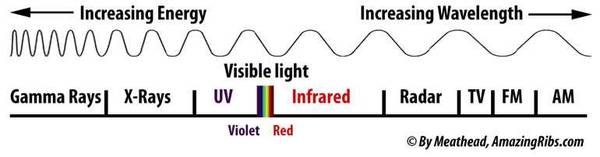
Now please keep in mind that radiation takes many forms and all are not scary like X-rays or gamma rays. Music arrives via sound waves, a benign, most welcome form of radiation. In this graphic of the electromagnetic spectrum you can see that radiation comes in the forms of waves, and the distance between the waves, the wavelengths, can be as big as a football field and smaller than the nucleus of an atom. The smaller the waves the more energy they pack.
IR is an intense form of light that is mostly invisible to the human eye. IR is energy emitted by from flames or glowing coals (or, if you are indoors, your broiler). This energy arrives at the speed of light and carries a real punch. That’s why food browns and cooks much faster when exposed to IR by sitting directly above the coals or flame, what we call direct energy. And the closer it is to the energy source, the more energy is imparted on the food. IR is a great way to get a sear on your steak if the energy source is strong enough and the steak is close enough.
It’s a tough concept to digest. Think of it this way: When you stand in your back yard, whether you are in the sun or the shade, the air temp is the same. Air temp is measured in the shade because the sun’s IR energy heats the thermometer and it delivers the temp of the instrument itself, not the temp of the air. That’s why it is misleading to take the air temp in the direct heat zone of a grill.
Max Good, Director of Equipment Reviews at AmazingRibs.com elaborates: “The sun radiates intense infrared (IR) waves that travel great distances at the speed of light without dissipating until they contact solid matter, like your skin, which it warms. The sun also radiates other waves, like ultraviolet (UV), which cause sunburn as well as heat. So all IR heat is radiant, but not all radiant heat is IR.”
As another example Good says “Consider the IR heaters in warehouses or bus stops. They are often suspended from the ceiling, but one can easily feel the heat waves standing beneath them. If these same devices were simply conventional gas burners, most of the heat would rise upward. Convection heat can be dissipated easily by air currents and distance while IR gets through.”
The AmazingRibs.com science advisor Prof. Greg Blonder adds “An oven set on broil is a radiant heater, with much of the heat is in the near IR, the almost visible section of IR. In principle, a pure IR heater would not be visible to the naked eye, since our eyes can’t respond that far out in the light spectrum. In practice, an IR heater and charcoal glow a dull red.”
Because IR packs such a wallop, the surface of the food can burn rapidly, before the interior cooks, so IR must be used judiciously, usually just to brown the surface. This is where a lot of newbies go wrong. They put the food directly over hot coals of gas burners and when the exteriors are really dark or starting to burn they take the meat off. But it is raw in the center, and undercooked poultry is dangerous.
Direct radiant energy is best used when you want to brown a surface and still get grill flavor. You have to watch the food carefully and turn it often.
Direct cooking outdoors is very much like broiling indoors. Both are direct radiant heat cooking. Indoor ovens usually have “Hi-Med-Lo” for broilers but not temperature gauges. That’s because the energy reaching the food changes a lot as you move closer or further from the flame. And every broiler is different because every broiler puts out different amounts of energy. That’s why a pro knows you have to watch a broiler closely, just like you have to watch a grill when using direct heat.
This is why I just shrug when people brag that their fancy new ceramic Big Blue Spaceship can get up to 900°F. That is not a good way to measure IR. That is not a good way to measure IR. You can’t measure IR in degrees F or C. IR is best measured in calories or joules.
A calorie with a lowercase c is the amount of energy needed to raise the temperature of 1 gram of water 1 degree Celsius. A joule is about 4.2 calories. The Calories with a capital C that you see on a food package are actually kilocalories, or 1,000 calories. A Calorie (kcal) is the amount of energy needed to raise the temperature of 1 kilogram of water 1 degree Celsius. In other words the Calories on a food package are 1,000 times larger than the calories used in chemistry and physics.
Originally, to determine the number of Calories in a food the food was placed in a calorimeter, a sealed container surrounded by water. The food was burned and the rise in water temperature was measured. Nowadays it is all done by computer because the Calories in the components of most foods is known.
That’s why, when talking about cooking in the not-hot indirect convection heat zone I specify a temperature like 225°F, but in the direct heat infrared zone measuring temperature is hard to do, not an accurate measure of the energy, and because every grill has different capabilities, especially gas grills, I specify a “Warp” number. Give ‘er all she’s got, Scottie, is Warp 10, and Warp 5 is about the middle of your dial.
The visible light produced by combustion is also an indicator of how efficient the burn is. During combustion the fuel produces gases that become part of the smoke. If all the gases burn completely, the flame is blue, as in a gas grill that has the proper mix of gas and oxygen. If the gases don’t burn completely, the flame is yellow or orange. If unburned gases escape, they cool and turn into part of the smoke.
In the past few years a number of gas grills have been touting their superiority because they use infrared heat or they have an IR sear burner or sizzle zone designed to generate high heat for getting a dark sear on meat. This is an attempt by gas grill manufacturers to close the gap with charcoal grills. In an IR gas grill a plate of special glass, ceramics, or metal absorbs the heat and light from the flame and re-radiates it to the food in the form of IR.
Dr. Blonder explains: “The atoms in the plate begin to oscillate. At low temps almost all of the energy is stored as vibrations in the plate. As the plate heats up, the rapidly vibrating atoms cause it to emit IR. Think of it (very loosely) as a teapot on a stove. At low temps most of the energy is confined the simmering liquid. At higher temps there is so much vibration that the water evaporates, forming steam and carrying away energy as a scalding hot jet.” At the top of the page is an IR sizzle burner under cast iron grill grates on a Napoleon Gas Grill.
Is IR heating better? Dr. Blonder says “IR energy is delivered faster than convection, but slower than conduction. So it can brown a little more effectively than a conventional grill but not as fast as a hot pan or grill grates. There are fewer hot spots from IR cookers. On the other hand, you can get thermal runaway with IR. If one section of the meat is dark, it absorbs more IR than the lighter sections. It browns, becomes darker still, and eventually burns.” And real IR burners are not good at achieving low temperatures.
I recommend gas grills with IR burner sections, especially them for getting dark sears on steaks and other meats when the browning of the surface creates thousands of new flavor compounds.
How your grills work
The illustration below shows how different outdoor cookers work.
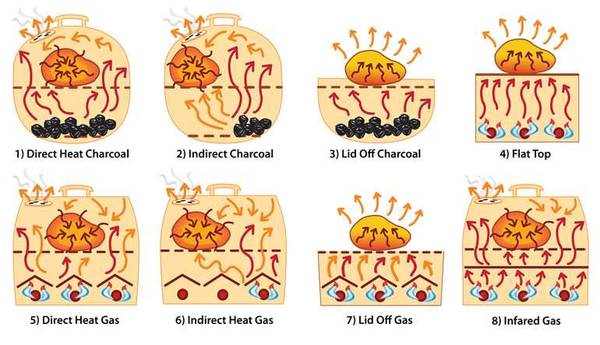
1) Direct Heat Charcoal or Wood. Charcoal produces radiant heat in the bottom. The grates absorb heat and produce conduction heat on parts of the surface of the food making grill marks. The lid reflects mostly convection heat. The exterior of the food absorbs direct heat from below, converts it to conduction heat, and it moves to the center of the food.
2) Indirect Heat Charcoal or Wood. Charcoal off to the side produces both convection and radiant heat. The grates absorb heat and produce conduction heat on surface of the food making grill marks. The exterior of the food absorbs indirect convection heat from all sides, converts it to conduction heat, and it moves to the center of the food.
3) Lid Off Charcoal or Wood. Charcoal produces radiant heat. The grates absorb heat and produce conduction heat on surface of the food making grill marks. The exterior of the food absorbs radiant heat from below only. The bottom of the food converts the absorbed energy into conduction heat, and it moves to the center of the food. Without a lid, heat escapes from the food upward, cooling its top.
4) Flat Top. Gas or charcoal produce radiant heat in the fire box. Solid griddle under the food absorbs heat and produces conduction heat on surface of the food. The bottom of the food absorbs conduction heat, browns where it is in contact with the surface, and conducts heat towards the top of the food. Without a lid, heat escapes from the food upward, cooling its top.
5) Direct Heat Gas. Burners produce radiant heat that heats metal drip protector bars, lava rocks, or ceramic briquets. They absorb heat and produce radiant and convection heat. The grates absorb heat and produce conduction heat on surface of the food. The exterior of the food converts all this incoming energy to conduction heat, and it moves to the center of the food.
6) Indirect Heat Gas. Burners off to the side produce radiant heat that heats metal drip protector bars, lava rocks, or ceramic briquets. They absorb heat and produce radiant and convection heat. The grates absorb heat and produce conduction heat on surface of the food. The exterior of the food absorbs indirect convection from all sides. The exterior of the food converts the energy to conduction heat and it moves to the center of the food.
7) Lid Off Gas. Burners produce radiant heat that heats metal drip protector bars, lava rocks, or ceramic briquets. They absorb heat and produce radiant and convection heat. The grates absorb heat and produce conduction heat on surface of the food. The exterior of the food converts the energy to conduction heat and it moves towards the top of the food. Without a lid, radiant heat escapes from the food upward, cooling its top.
8) Infrared Gas. Burners produce radiant heat that in turn heats a special plate. It absorbs heat and emits it as radiant heat in the infrared part of the spectrum. Grates absorb heat and produce conduction heat where they contact the surface of the food. The bottom of the food absorbs the radiant heat and converts it to conduction heat and moves it to the center of the food. The top of the food is heated by indirect convection heat bounced from the lid.
How a smoker works
Smoke is the perfume, the sexy scent that makes us stand outdoors on cold rainy nights nurturing the rib cage of a hog as if we were delivering a child. Smoke is the indescribable difference between the crockpot pulled pork they serve at the ballpark and the hand crafted sandwich that gets you a standing ovation. Smoke is what makes brisket simmered in a flavorful stew run and hide its head when it encounters the stuff that Texans drive 50 miles for, just for lunch.
But not just any smoke will do. Nobody wants their turkey tasting like cigarettes. So there is an art to getting the right smoke. You can make superb smoked foods on just about any grill with a cover, even a gas grill. But adapting a grill for smoking is like adapting the family car for racing. The better solution is to buy a race car.
With a high quality modern barbecue smoker, and a little practice, you can make food better than the ribs, brisket, and other faves holding in the warming ovens of restaurants. With a good modern smoker you no longer need to hover over your machine for five hours, constantly monitoring the temp, fiddling with the dampers, shoveling coal, adding wood, and spritzing your meat with a mist of secret moisturizer. With a good smoker you can get your food on the table on time and without fear that it is over-cooked (an expensive waste) or under-cooked (dangerous).
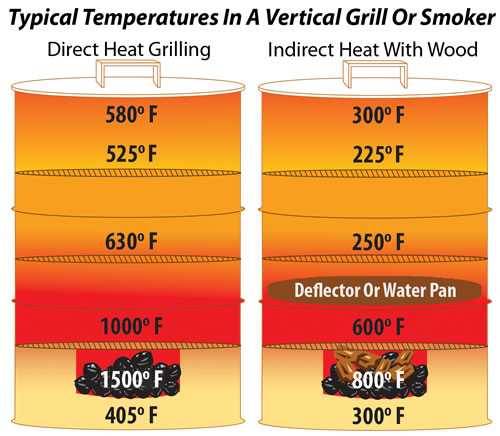
Most smokers have an indirect heat source with a deflector or a water pan that protects the food from searing infrared radiation and cooks with convection. They generate smoke by burning wood. A few use wood for both heat and smoke, but most use charcoal, gas, or electricity for heat, and wood for smoke.
Master of the flame and keeper of time
Good cooking is finding the proper combination of heat and time. The higher the heat, the less time needed. Lower the heat, you need more time. But you just can’t set your cooker for Warp 10 on every food. Some demand low and slow. Read more about cooking temperatures here.

Your indoor oven has three basic components to control heat and time: A heat source, a thermostat, and a timer. Your outdoor oven, and an oven is exactly what your grill or smoker is, has a heat source, but only a few have a thermostat or a timer. And that’s why cooking outdoors is much more difficult than indoors. Tell that to your significant other (and sleep on the couch).
To make good food outdoors you need to become master of the flame and keeper of time. Many grills come with a thermometer, but they are usually crap. The thermometer on my gas grill is often off by 50°F! You absolutely positively must have a good oven thermometer.
In addition to knowing what temp your cooker is, you need to know what temperature your meat is. Cooking without a meat thermometer is like driving without a speedometer. You might think you’re under the limit, but try explaining that to the judge. You might think the meat is ready, but try explaining that to your guests when it is dry enough that they ask for a glass of saliva to help them swallow. Here is a Food Temperature Guide, and here’s everything you need to know about thermometers with links to good ones you can buy.
Some of the better digital thermometers have timers. If yours does not, get in the habit of wearing a watch when you cook and write down what time you put the meat into the heat in a cooking log. Some of the old timey barbecue artisans say you can learn to cook by sight, smell, and feel. That’s just plain BS. Once you know how to cook you can rely more and more on your senses, but until you are ready to call yourself a top chef, get a thermometer, wear a watch, and keep a log. I do.








Why We Require You To Sign In Before You Can Post Comments
Before you can post a comment or question you must sign into our commenting partner, Disqus. This is helps make sure everyone hanging around the grill is civil. We do not tolerate nastiness, racism, porn, inappropriate language, or attacks on others. All comments are the property of AmazingRibs.com and we reserve the right to quote them, edit them, delete them, and block people from making future comments.
Please leave comments and questions on the page devoted to the same subject so others can see them and our answers when they are reading about that subject. You must enable JavaScript to use the comments section, and you must accept cookies to post comments. Note that the software that runs Disqus is different from the Pitmaster Club so members need to sign into that separately.
Moderators